Atoms are constantly moving — yet DFT freezes everything at 0 K. Any accurate description of a "spontaneous" reaction, formation, or synthesis requires free energy, not internal energy.
Free Energy in Heterogeneous Catalysis
From EDFT to G(T, P, U)
Table of Contents
Why Free Energy Matters
The problem with the potential energy diagram
For Realistic Modeling

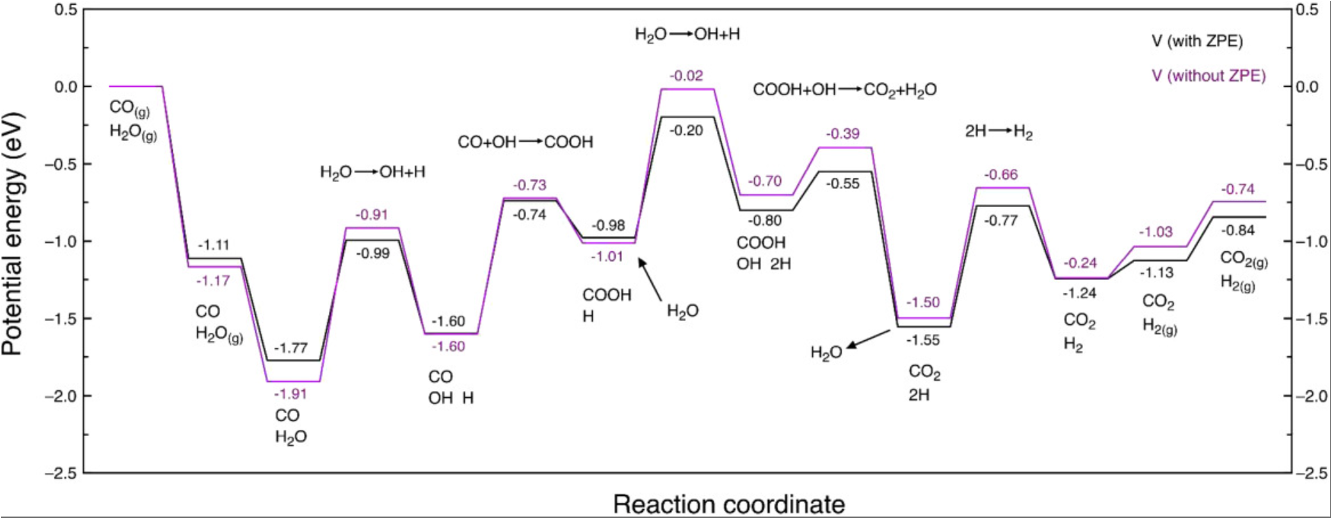
When PED Gives the Wrong Answer
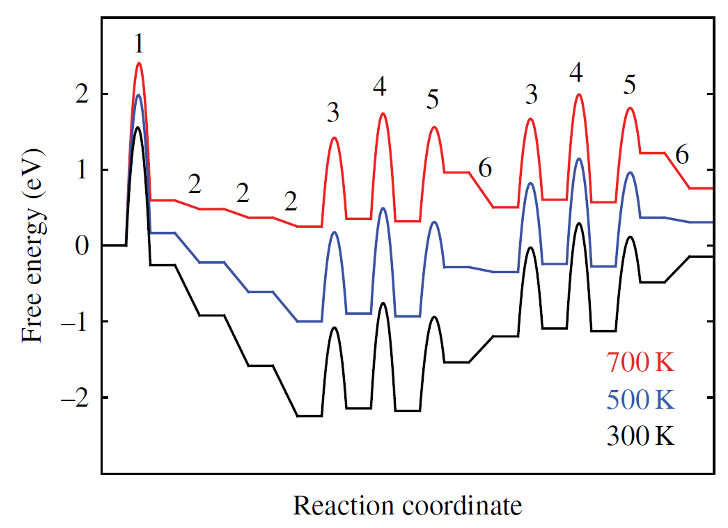
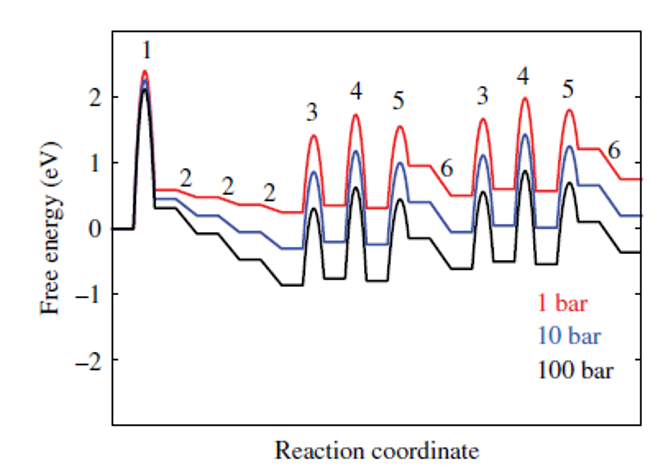
NH3 Synthesis — sign reversal (700 K)
CO on Pt — entropy dominates
- Equilibrium coverage θi(T, P)
- TOF via energy span model
- Selectivity: products set by ΔG, not ΔE
- TST: $k = \frac{k_B T}{h} e^{-\Delta G_{\text{TS}}/k_B T}$
Activity and Selectivity Are Sensitive to Free Energy
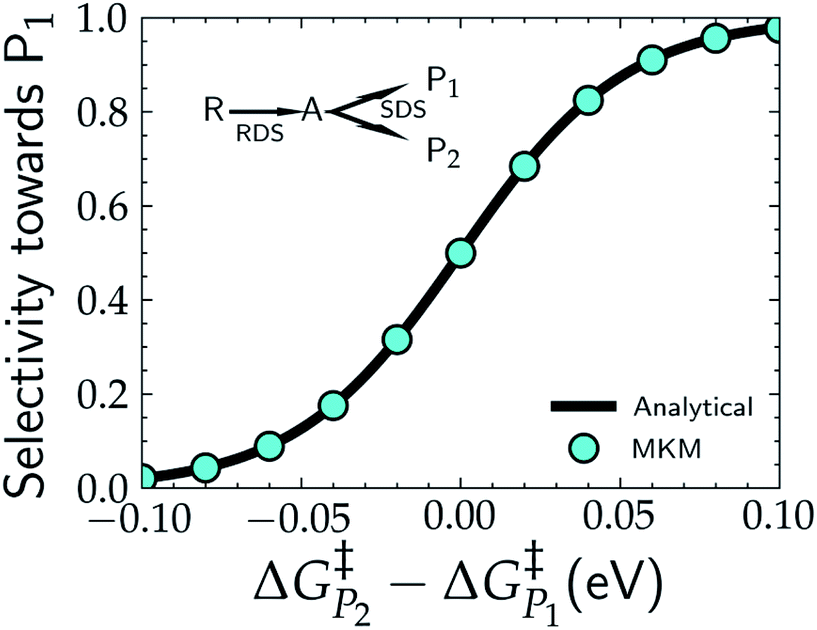
Selectivity switches sharply within $\pm 0.05$ eV of $\Delta G_{P_2}^{\ddagger} - \Delta G_{P_1}^{\ddagger}$
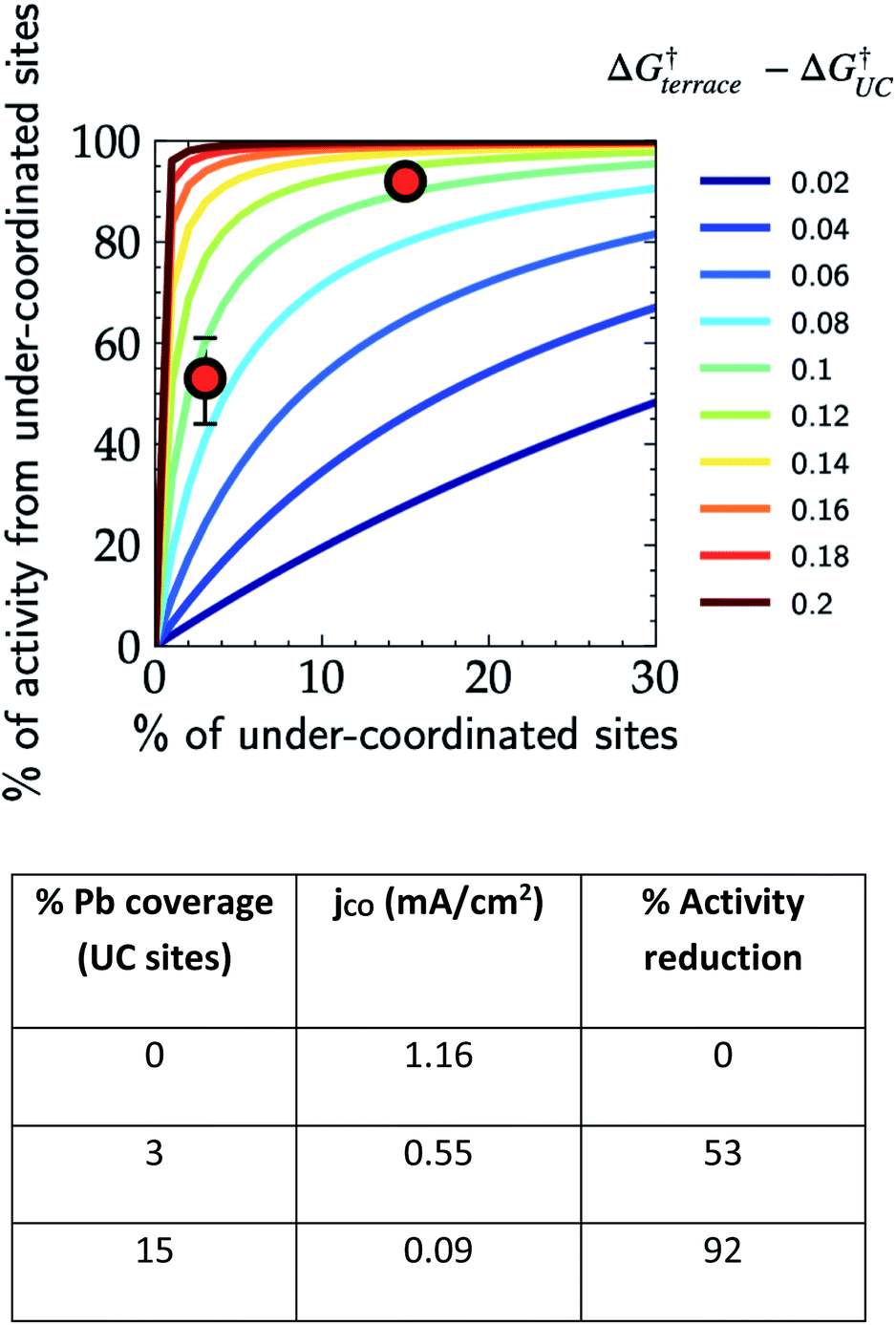
Contribution of under-coordinated sites to overall activity vs. their abundance and $\Delta G_{\text{terrace}}^{\dagger} - \Delta G_{\text{UC}}^{\dagger}$. Red markers: Pb-UPD on Au(111) for CO2RR at −0.6 V vs. RHE.
Figures: Govindarajan, Kastlunger, Heenen & Chan, Chem. Sci. 2022, 13, 14–26. DOI: 10.1039/D1SC04775B
ZPE and Entropy
The quantum ground state and the thermal ensemble
Zero Point Energy: Physics First
$E_{\min} = \tfrac{1}{2}h\nu$ above the potential well floor — Heisenberg uncertainty forbids a stationary particle
ZPE formula
- $\text{ZPE} = \sum \tfrac{1}{2}h\nu_i$ over all real vibrational modes
- N–H, O–H (above 3000 cm⁻¹): contribute 0.2–0.5 eV
- Low-frequency frustrated modes (below 200 cm⁻¹): negligible
- Only the change in ZPE between states matters (ΔZPE)
Quantitative warning
- NH3 synthesis: 0.83 eV too exothermic without ZPE (Norskov Table 2.1)
- H-transfer reactions: always include ZPE
- Safe to ignore: heavy adsorbates with no H transfer (ΔZPE below 0.05 eV)
Vibrational Entropy and Low-Frequency Corrections
Entropy: Five Practical Approaches
Set $\Delta S_{\text{ads}} = 0$
Justified for surface-to-surface steps where entropy difference is below 0.05 eV. Fast and defensible for large-scale screening. (Norskov CHE 2004)
$S_{\text{ads}} = 0.70 \cdot S_{\text{gas}}$
Campbell and Sellers, JACS 2012. Factor 0.5–0.9 depending on binding strength. Appropriate for mobile physisorbed or weakly bound adsorbates.
Full harmonic partition function (ASE / VASPKIT)
T·S error typically 30–50% of surface mode contribution. Chemisorbed: T·S ~ 0.05–0.15 eV. Physisorbed: T·S ~ 0.3–0.5 eV with much larger error.
Ab initio MD (AIMD)
Sample the full potential energy surface at finite $T$ via Born-Oppenheimer or Car-Parrinello MD. Captures anharmonicity and diffusion naturally. Cost: ~100–1000× static DFT; limited to ps timescales and small cells.
MLIP-based MD
Machine-learned interatomic potentials (MACE, NequIP, CHGNet, etc.) trained on DFT data. Near-DFT accuracy at classical MD cost — enables ns timescales and large supercells for converged free energy sampling via thermodynamic integration or metadynamics.
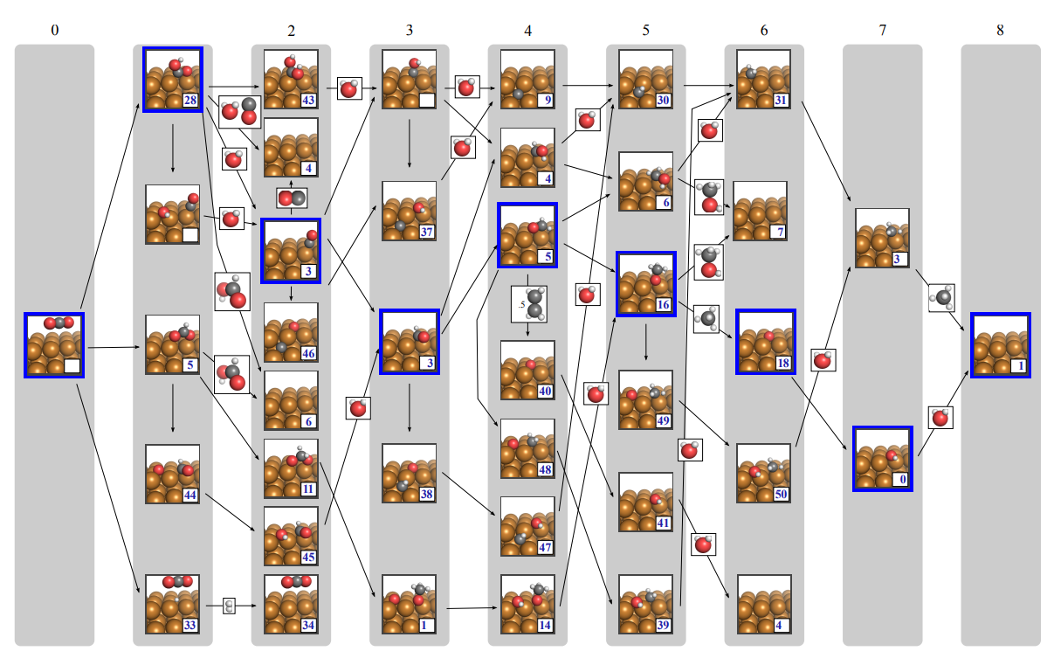
Configurational Entropy of Adsorbates
$N$ adsorbates occupying $N_0$ surface sites ($\theta = N/N_0$) give rise to a configurational entropy that depends on coverage. * denotes a vacant site.
When it matters
- Microkinetic models solving $\theta$ self-consistently (e.g. CatMAP)
- Phase diagrams comparing surfaces at different coverages
- At $\theta = 0.5$: $T \cdot s_{\text{config}} \sim 0.018$ eV (300 K), 0.059 eV (1000 K)
When safe to ignore
- Comparing states at the same coverage — $S_{\text{config}}$ cancels
- Dilute limit ($\theta \to 0$) or saturated ($\theta \to 1$)
- Most CHE free energy diagrams omit this deliberately
Gas Phase Thermodynamic Examples
Where entropy and enthalpy compete
CO2 Reduction: Why Product Choice Changes Everything
Equilibrium potentials at 298 K, pH 0 vs. SHE
CO2 → CO (2e⁻)
TΔS ≈ 0 — same gas moles on both sides; entropy gives no relief
CO2 → CH4 (8e⁻)
Large favorable ΔH overwhelms entropy penalty; thermodynamically easiest per electron
Gas-Phase Reference State and μ(T, P)
Two routes to T-dependent terms
- ASE IdealGasThermo — uses DFT vibrational frequencies
- NIST-JANAF tables — tabulated $[H(T) - H(298)]$ and $S(T)$ directly from experiment; no DFT frequencies needed. Covers most common gas molecules (H2, O2, H2O, CO2, NH3, CH4, etc.)
Worked example for H2O
At 298.15 K, 1 bar
Pressure term: $k_BT\ln(1/1) = 0$ eV
$\mu = E_{\text{DFT}} + \text{ZPE} + H_{\text{thermal}}(298) - 298 \cdot S(298)$
At 500 K, 0.01 bar
Pressure correction: $0.04313\ln(0.01) = -0.20$ eV
$\mu$ shifts by $-0.20$ eV relative to 1 bar
Phase Diagrams
Which state has the lowest G under reaction conditions?
Phase Diagram from First Principles
A phase diagram maps which state has the lowest G as a function of T, P, pH, or U. Stability boundary: $\Delta G_{AB} = 0$
Construction
- Write $G(\text{variable})$ for each competing phase
- Lowest $G$ at each condition → stable phase
- Intersections define stability boundaries
- Variables: $T, P(\text{O}_2)$; $U, \text{pH}$; $\mu_C, \mu_N$
Example: PdOx/CeO2 CH4 Oxidation
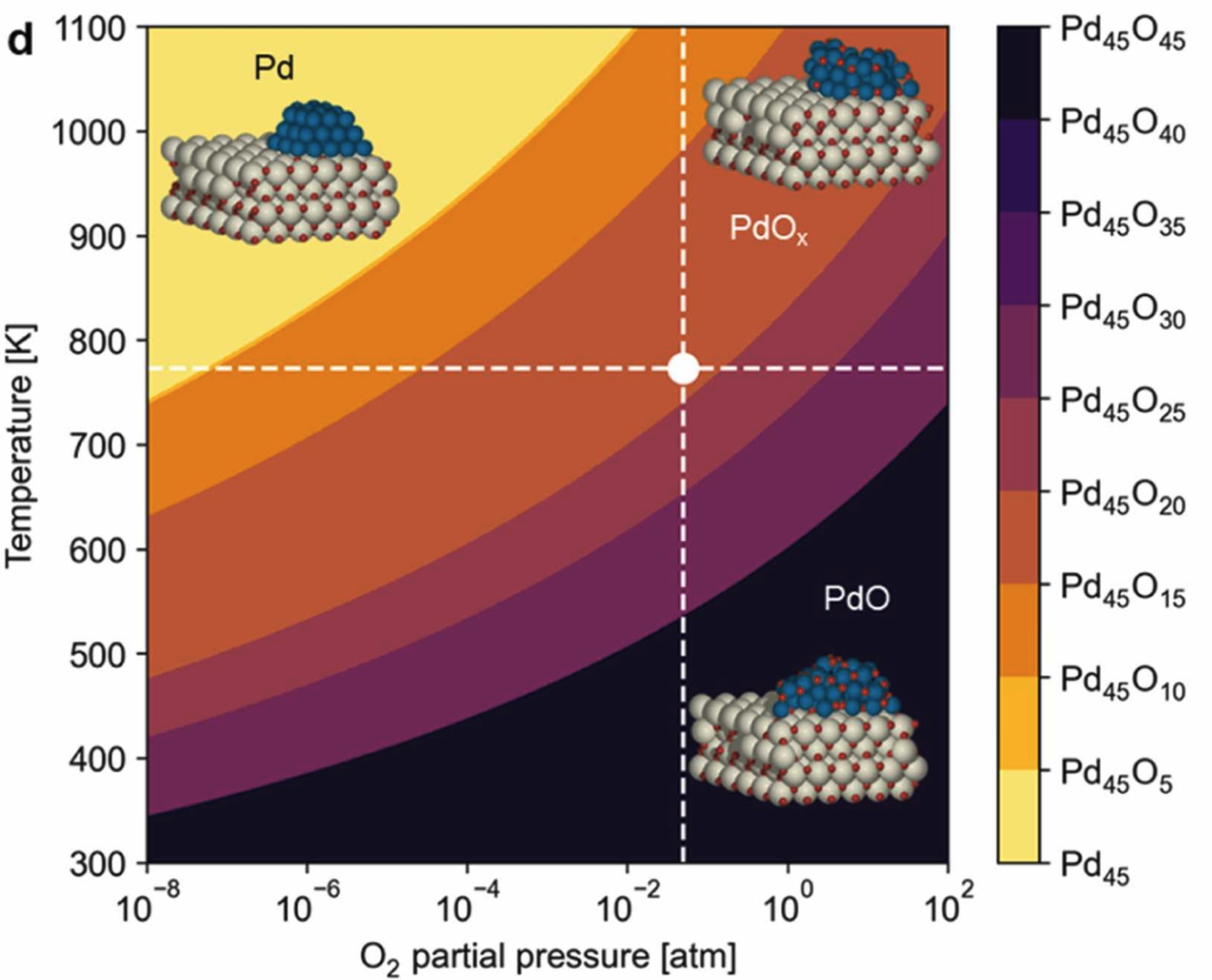
Ryu, Choung et al. Appl. Catal. B 379 (2025) 125672
$\Delta G_{\text{ox}}(T, P_{\text{O}_2}) = \Delta G_f^{\circ}(\text{PdO}) + \tfrac{1}{2}RT\ln(P_{\text{O}_2}/P_{\text{ref}})$
- Defect-rich CeO2 stabilizes partially reduced PdOx via Ce3+ electron donation — shifts boundary to higher T
- Active state at phase boundary; dynamic redox pulse transiently crosses it
Surface Pourbaix Diagram
Construction
- Write $G(U, \text{pH})$ for each surface state. Slopes set by $n$ (electrons transferred).
- Lowest line at each $U$ is stable. Intersections → phase boundaries.
- Sweep pH 0–14 for 2D map. pH shifts by 0.0592 V/pH at 298 K.
Example: Fe(OH)x/Pt ORR
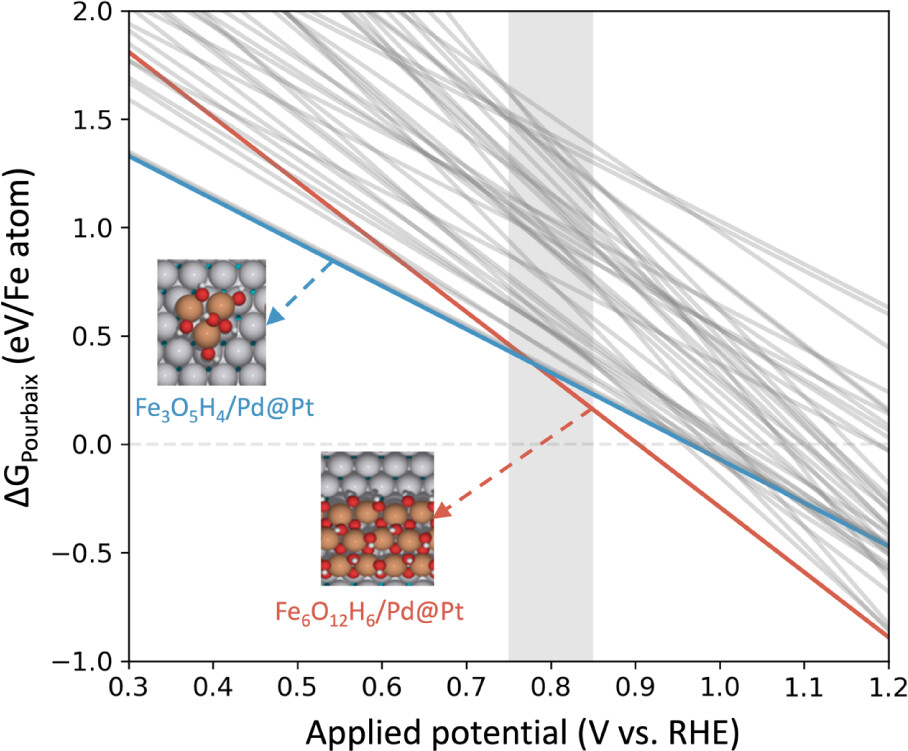
Maiti, Choung et al. ACS Appl. Mater. Interfaces 17 (2025) 40517
- At ORR conditions (0.7–0.9 V vs. RHE): Fe3+(OH)3 or FeOOH is stable — not metallic Fe
- Pt substrate shifts boundaries vs. bulk Fe Pourbaix via charge transfer
- Engineer interface to pin Fe3+ oxidation state at operating potential
Example 3: Rh SAC Stability on Fe-Ce Oxide
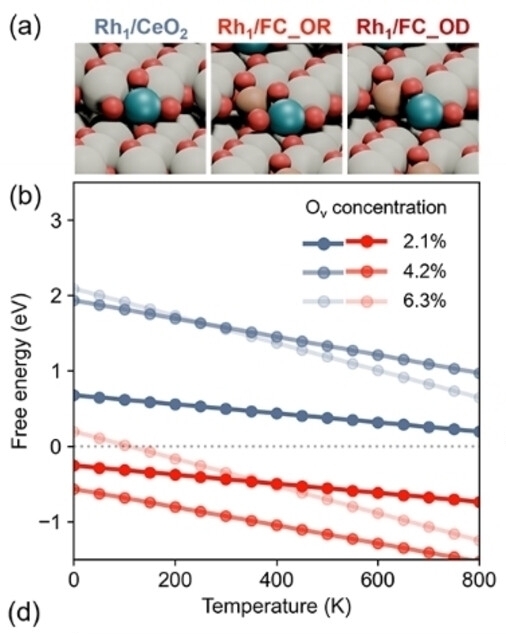
Kim, Choung et al. Angew. Chem. Int. Ed. 64 (2025) e202421218
Does Rh+ SAC remain stable vs. sintering to Rh⁰ under reaction conditions?
Competing states: Rh+/defect, Rh3+/stoichiometric, Rh⁰ cluster, dissolved Rhn+
- Ce3+ defects bind Rh+ ~0.5–1.0 eV more strongly than stoichiometric sites
- Defect-rich Fe-Ce oxide expands the Rh+ SAC stability window to lower P(O2) at high T
- Phase diagram directly predicts sintering resistance — confirmed by durability experiments
- Operating point inside SAC-stable region only for defect-rich support
Electrochemistry and CHE
The computational hydrogen electrode
Free Energy in Electrochemistry: The CHE Model
Norskov et al. (2004): at U = 0 V vs. RHE, G(H⁺ + e⁻) = ½G(H2). Shifting U by −eU shifts every electron transfer step.
ORR Worked Example: Step-by-Step Free Energy Diagram
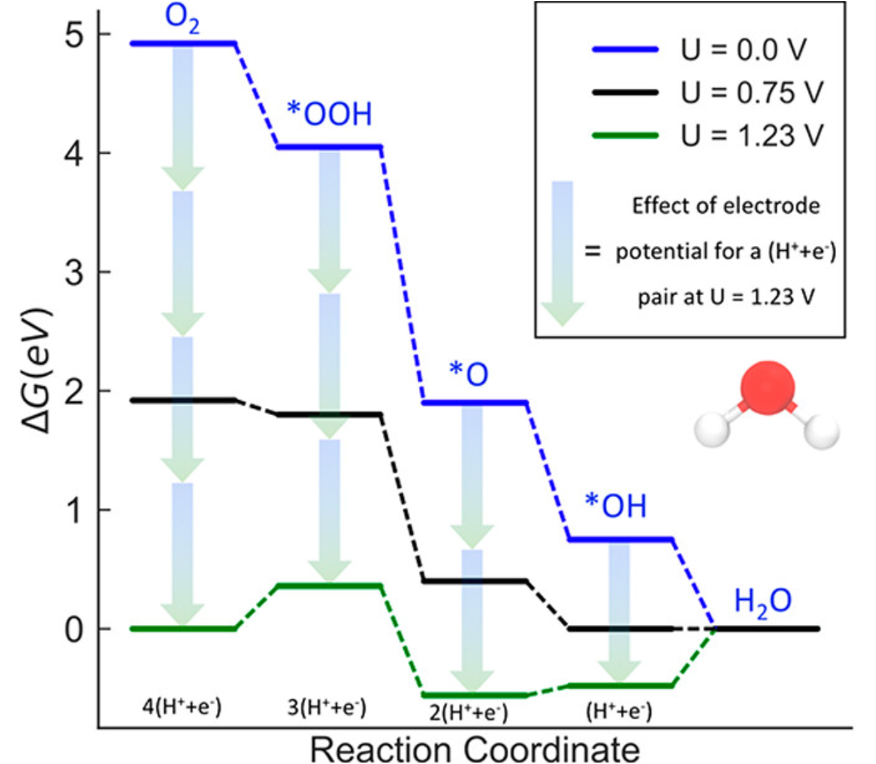
O2 + 4(H⁺ + e⁻) → 2H2O | 4-step associative mechanism on Pt(111)
How Wrong Can a Free Energy Calculation Be?
Error tiers: functional, model, thermal, and site selection
Error Tier 1: DFT Functional
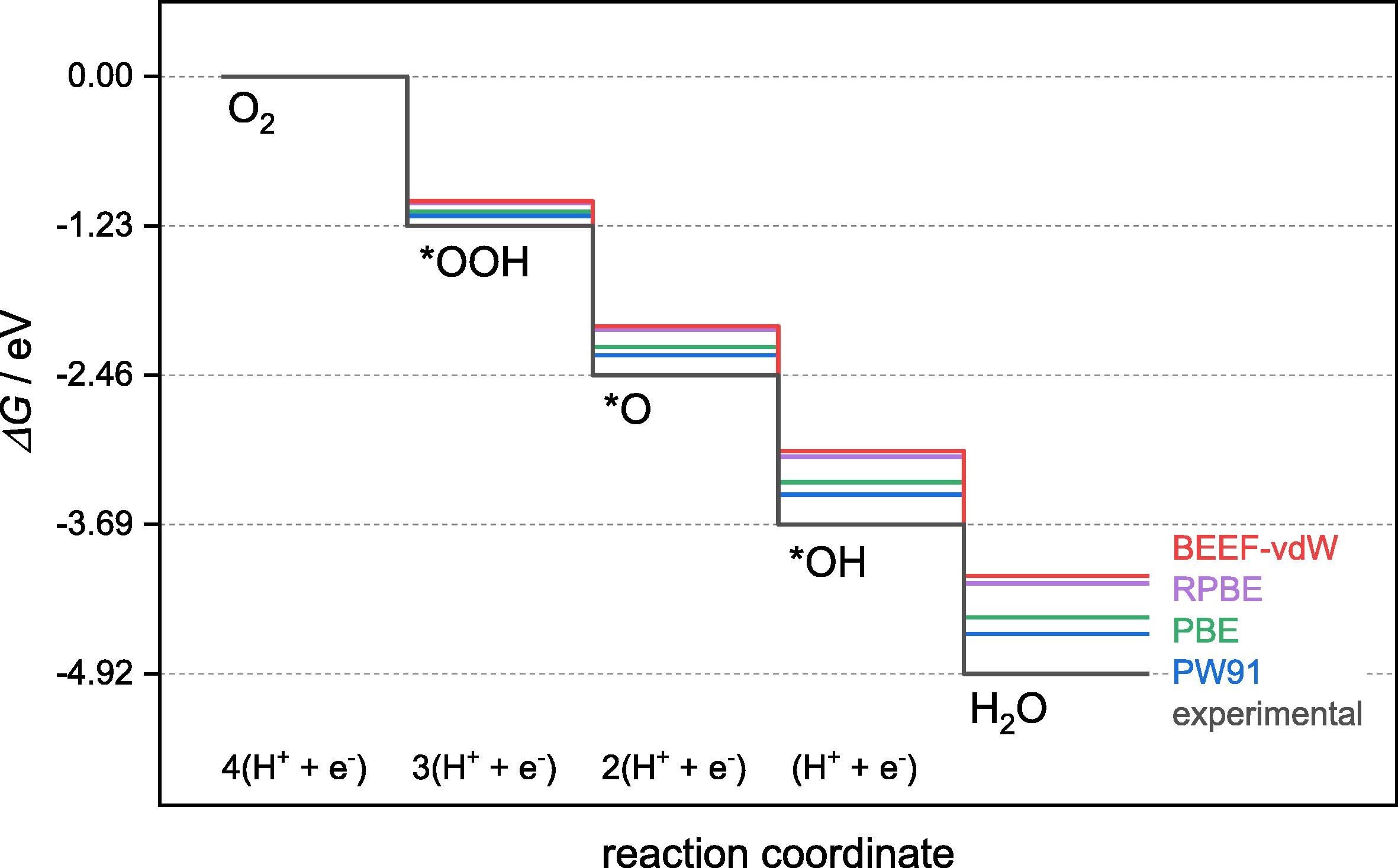
Sargeant, Illas, Rodríguez & Calle-Vallejo, J. Electroanal. Chem. 896 (2021) 115178
GGA-PBE overbinds O-containing adsorbates
Systematic error of 0.2-0.4 eV for O*, OH*, OOH* on metals. Not random — correlated across similar adsorbates, so volcano plots still rank catalysts correctly. Absolute overpotentials can be wrong by 0.2-0.4 V.
When to worry
- Absolute adsorption energies vs. experiment
- Comparing different functional groups (O vs. N adsorbates)
- Oxides — GGA+U or HSE06 required
When errors cancel
- Relative energies within the same adsorbate family
- Volcano plot ordering (errors are correlated)
- Scaling relation slopes (largely functional-independent)
Error Tier 2: Model and Setup
Error Tier 3: Which Site Did You Model?

Govindarajan, Kastlunger, Heenen & Chan, Chem. Sci. 2022, 13, 14–26
- DFT screening typically picks one binding site. If the chosen site is not the kinetically dominant one, the prediction fails regardless of numerical accuracy.
- Au(111) Pb-UPD contour analysis (Nitopi et al., Chem. Sci. 2022): 80-120 meV barrier difference between terrace and under-coordinated sites translates to 20x-100x higher TOF at UC sites via Arrhenius.
- Co oxide OER: minority defect sites show 100x higher TOF than average Co surface sites (Frei time-resolved FTIR; Plaisance microkinetic modeling). Dominant active site changes with applied potential.
- A 0.10 eV barrier difference is smaller than typical DFT error bars, yet it shifts which site controls the rate by 1-2 orders of magnitude. Site counting (Pb-UPD, redox peak integration, CO/CN probes) is essential.
- Even perfect DFT kinetics can be masked by mass transport limitations in experiment (low solubility of H2, O2, CO2).
FAQ
Common questions about free energy calculations
When Can I Safely Ignore ZPE and Entropy?
- Surface-to-surface steps with no H transfer and similar reactant/product masses — ΔG ~ ΔE within ~0.05 eV
- Screening large catalyst libraries (hundreds of materials) where trends matter more than absolute values
- Comparing within the same adsorbate family on similar metals — errors cancel in differences
- For gas-phase reactions at T above 400 K: entropy is never negligible — use NIST thermochemical tables
- For electrochemistry: CHE handles gas-phase T·S via H2 reference, but surface T·S still matters for rate-limiting steps
Which Entropy Approach Should I Use?
Surface-to-surface steps; fast screening
ΔS ~ 0 when both reactant and product are chemisorbed. T*ΔS below 0.05 eV. Default for large-scale DFT + CHE screening.
Physisorbed or weakly bound intermediates
Mobile adsorbates (large molecules on surfaces, physisorption). Campbell factor 0.5-0.9 depending on binding energy. Quick estimate.
Rate-limiting steps, validation, publication
Use ASE HarmonicThermo or VASPKIT 501. Report quasi-RRHO correction for modes below 100 cm⁻¹. Required for quantitative overpotential claims.
What Are the Limitations of the CHE Model?
- No kinetic barriers — CHE is purely thermodynamic; actual barriers may differ by 0.3-0.8 eV
- No explicit solvent — implicit solvation can shift ΔG by 0.1-0.3 eV for polar intermediates
- No coverage dependence — lateral interactions shift onset potential by 0.1-0.2 V
- No Frumkin corrections — assumes ideal double-layer; fails at high overpotential
- Not for proton transfer steps involving Grotthuss hopping — need explicit water molecules
How Do I Set Up Correct Reference States?
- Always reference all energies to the same chemical potentials: H → ½G(H2), O → G(H2O) − G(H2), C → G(CO2) − G(O2)
- Never mix DFT-computed gas-phase G with experimental enthalpies in the same pathway
- Supercell size affects absolute surface energy but not ΔG for chemisorption if the slab is converged
- For phase diagrams: set μO(T, P) = ½G(O2) + ΔμO(T, P) from NIST-JANAF or phonon calculation
How Should I Estimate and Report Free Energy Uncertainty?
- DFT functional: +/- 0.2-0.4 eV for O* family on metals (PBE); cite a systematic benchmark for your specific system
- Thermal corrections: +/- 0.05 eV for chemisorbed at 300 K; +/- 0.15 eV for physisorbed or T above 700 K
- Model setup: converge slab thickness, k-mesh, ENCUT systematically and report convergence criteria explicitly
- Propagate errors: use linear error propagation for multi-step pathways; $\sigma_{\text{total}} = \sqrt{\sum \sigma_i^2}$
EDFT is a photograph. G(T,P) is a film.
Free energy tells the full story of what actually happens
Every correction matters
Know what you are ignoring, and why
The phase diagram does not lie
Compute boldly. Correct carefully. Interpret honestly.